Techniques
Behavioural studies
Students in the lab have used a variety of behavioural tests in their projects, such as preference/avoidance olfactory assays, multi-sensory integration assays, appetitive or aversive conditioning, maze training, and manipulations of motivation. These tests allow us to investigate the evolution of learning and cognition in vertebrates. We have conducted behavioural experiments with amphibians, fishes, and even a reptile species (leopard gecko). The picture shows a fire-bellied toad attending to prey stimuli displayed on a computer screen, part of a visual discrimination learning experiment.
Descriptive neuroanatomy and histology
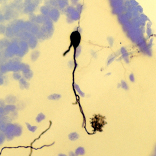
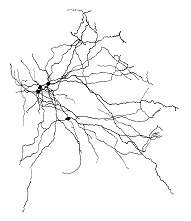
We have performed tract tracing of brain pathways and intracellular labelling of single/few neurons in in vitro brain preparations. A fascinating feature of the brains of anamniote vertebrates is that you can keep this organ alive in physiological solution for an extended period of time, allowing for experiments with neurotracer substances directly on the isolated brain. The detection of tracer substances on brain tissue can be done using either light or fluorescence microscopy. The micrograph on the left shows the cell body and processes of a mitral cell contained within one brain section of the salamander accessory olfactory bulb labelled by intracellular injection of biocytin. The figure on the right shows the reconstruction of a cluster of thalamic neurons in the fire-bellied toad. We have an imaging system equipped with the software Neurolucida (MBF Bioscience), which allows reconstruction of neurons in 3D-like fashion. The lab is equipped for histology work using thick sections cut on a vibrating microtome or paraffin-embedded tissues.
Functional neuroanatomy
Methods that indirectly measure brain activity enable the study of whole brain responses in one experiment and could help determine which brain regions are engaged by the conditioning procedures established in our behavioural studies. We are trying to develop in situ hybridization against immediate-early genes or other genes engaged by behavioural tasks in the fire-bellied toad in collaboration with the Heyland lab. We also have an interest in testing the effectiveness of recently developed methods (e.g. Knight et al. 2012 Cell 151:1126-1137) of functional neuroanatomy for investigations in anamniote vertebrates.
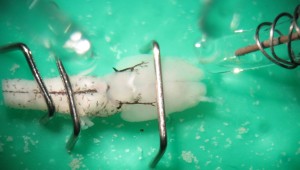
Electrophysiology
The lab has a sharp electrode intracellular electrophysiology system that can be used for intracellular labeling of neurons and larger tracer injections in isolated brain preparations. Mechanisms of sensory integration and potentiation in regions critical for behaviour can also be investigated in the in vitro brain preparation by artificial sensory stimulation, which is accomplished by stimulation of sensory nerves and brain regions that relay sensory information. The picture below shows a suction electrode (top right) used to stimulate electrically the right olfactory nerve branch in a salamander brain (viewed ventrally). The presence of a recording glass electrode aimed at the right caudal telencephalon can be seen on the top left in the picture.